Advancing the Fight Against Ovarian Cancer
The number of women diagnosed with ovarian cancer in Australia increased from 833 in 1982 to 1,266 in 2007 [1]. An estimated 1,488 women are expected to be diagnosed with ovarian cancer in Australian in 2015 [2]. 75% of women with ovarian cancer present at an advanced stage (stages III / IV) with disseminated abdominal disease and ascites.
Screening
Presently, there is no evidence that population screening for ovarian cancer is of any benefit in reducing mortality from this disease. Even with combined tumour markers and imaging, overall survival is not improved whilst intervention rates are increased where 10 laparotomies or laparoscopies need to be performed to find one cancer. However, screening may have a role in high-risk populations, such as a strong family history, gene mutation carriers, and Ashkenazi Jewish women.
Initial Cytoreductive Surgery
The amount of residual tumour deposits after surgical management of ovarian cancer has been consistently shown to predict survival. Each 10% increase in maximal cytoreduction was associated with a 5.5% increase in median survival time [3]. With 75% of this disease presenting at stage III / IV with ascites and abdominal deposits of tumour on bowel, liver, diaphragm, and other abdominal organs, surgery to achieve optimal debulking may be radical, extensive and potentially morbid.
This surgery, in addition to the removal of the ovaries and uterus, often involves a bowel resection, omentectomy, diaphragmatic, and subsegmental liver resection. Deciding on whether a patient is a candidate for such a surgical approach should be made after careful consideration of other co-morbidities and following discussion in a multidisciplinary team meeting. Optimal debulking to nil macroscopic visible disease in selected patients, remains the gold standard of care in ovarian cancer patients.
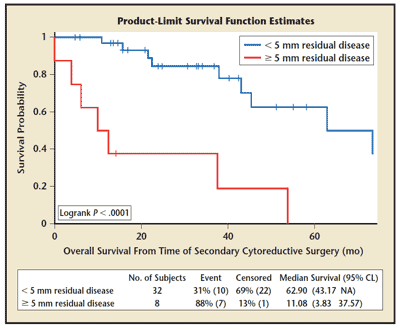
Figure 1. Overall survival of secondary debulking by amount of residual disease. CL, confidence limits; NA, not applicable. Reprinted from International Journal of Gynecology and Obstetrics, Vol. 108, Schorge JO et al, “Secondary cytoreductive surgery for recurrent platinum-sensitive ovarian cancer," pp. 123-127, Copyright 2010, with permission from Elsevier.
Intraperitoneal Chemotherapy
Adjuvant chemotherapy has traditionally been administered intravenously. However, recent randomized studies have shown that combination intravenous and intraperitoneal chemotherapy delivered via a port directly into the abdominal cavity achieves better progression free (18.3 vs 23.8 months P=0.05) and overall survival (49.7 vs 65.6 months P=0.03) [4].
This improvement in survival only applies when initial cytoreductive surgery has achieved an optimal debulking with no residual disease >1cm. Intraperitoneal chemotherapy was associated with more side effects at the time of treatment when compared to intravenous-only treatments, but the quality of life was equivalent at 12 months after treatment.
Neoadjuvant Chemotherapy with Interval Debulking
In patients whom we predict may not achieve optimal debulking at the initial cytoreductive surgery, neoadjuvant chemotherapy should be strongly considered. This approach involves three cycles of chemotherapy followed by interval debulking surgery; then, a completion of a further three cycles of chemotherapy – this approach has been shown to achieve similar progression free and overall survival with less morbidity [5].
Initial treatment with chemotherapy may reduce tumour volume to allow less radical surgery in order to achieve optimal debulking. Complete resection of all macroscopic disease, whether performed as primary treatment before chemotherapy or as interval debulking surgery after neoadjuvant chemotherapy, remains the objective because complete resection of all macroscopic disease (at primary or interval surgery) is the strongest independent variable in predicting overall survival.
Targeted Therapy
Any use of targeted therapy for ovarian cancer is still classified as experimental and is not standard practice. Whilst trials are still underway, evidence is slowly emerging that there are several classes of drugs that may be effective against ovarian cancer.
Preliminary studies of chemotherapy with angiogenesis (growth of blood vessels) inhibitor drugs such as bevacizumab (Avastin) have shown improved progression free survival but not overall survival from ovarian cancer [6]. PARP Inhibitors may have some utility in ovarian cancers related to the BRCA mutation [7].
On-going Follow-up
There is limited evidence to show that intensive follow-up of any gynaecological cancer improves outcomes. This is counter-intuitive as one would think that earlier detection of recurrences might improve outcome.
Certainly there is no role for frequent CT scans as radiation from CT scans may be harmful. A recent large multi-centre randomized control trial compared whether to follow-up ovarian cancer patients who are asymptomatic with blood tumour markers or not – it showed that patients followed up with routine blood tumour markers lived just as long as those who did not have frequent blood tests; however, those who did have the blood tests had a poorer quality of life [8]. Patients who had regular blood tests had their recurrences discovered earlier, but despite earlier re-treatments with chemotherapy, there was no survival advantage, more side effects and poorer quality of life.
Certainly there are some limitations to this study, including the non-utility of secondary debulking surgery. These patients were mostly re-treated with chemotherapy without surgery, and secondary optimal debulking surgery prior to further chemotherapy for recurrent disease has been shown to improve survival compared to chemotherapy alone.
| 1. | Australian Institute of Health and Welfare & Australasian Association of Cancer Registries 2010. Cancer in Australia: an overview, 2010. Cancer series no. 60. Cat. no. CAN 56. Canberra: AIHW |
|---|---|
| 2. | Australian Institute of Health and Welfare 2010. Gynaecological cancer projections 2010-2015. Cancer series no. 53. Cat. no. CAN 49. Canberra AIHW. |
| 3. | Bristow et al. Survival effect of maximal cytoreductive surgery for advanced ovarian carcinoma during the platinum era: a meta-analysis. Journal of clinical oncology : official journal of the American Society of Clinical Oncology (2002) vol. 20 (5) pp. 1248-59 |
| 4. | Armstrong et al. Intraperitoneal cisplatin and paclitaxel in ovarian cancer. New England Journal of Medicine (2006) |
| 5. | Vergote et al. Neoadjuvant chemotherapy or primary surgery in stage IIIC or IV ovarian cancer. The New England journal of medicine (2010) vol. 363 (10) pp. 943-53 |
| 6. | Burger RA, Brady MF, Bookman MA, et al. Phase III trial of bevacizumab in the primary treatment of advanced epithelial ovarian cancer, primary peritoneal cancer, or fallopian tube cancer: a Gynecologic Oncology Group study. J Clin Oncol 2010;18s:946s (LBA 1). |
| 7. | Chan and Mok. PARP inhibition in BRCA-mutated breast and ovarian cancers. Lancet (2010) vol. 376 (9737) pp. 211-3 |
| 8. | Rustin et al. Early versus delayed treatment of relapsed ovarian cancer (MRC OV05/EORTC 55955): a randomised trial. Lancet (2010) vol. 376 (9747) pp. 1155-63 |


























